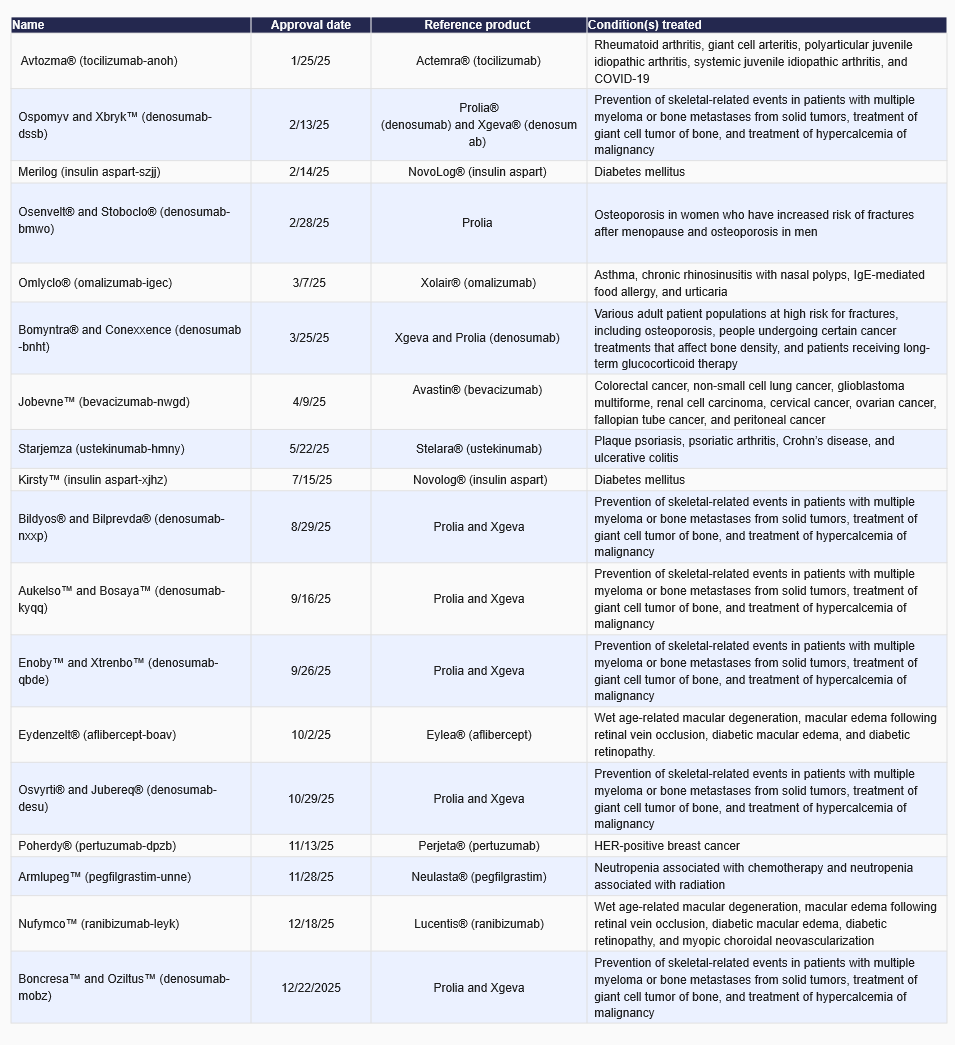
Summary
- The Food and Drug Administration (FDA) approved 18 biosimilars in 2025.
- February was the busiest month for approvals with three approved biosimilars.
What is a biosimilar?
According to the FDA, a biosimilar is a biologic that is highly similar to another biologic that has been approved by the FDA. A biosimilar can’t have any clinically meaningful differences from its original biologic (often referred to as the reference product) and must have the same route of administration, the same strength/dosage form, and the same potential side effects.
An interchangeable biosimilar has to meet additional requirements and can be substituted for the original product without consulting the prescriber. A non-interchangeable biosimilar requires a specific prescription from a doctor.
Biosimilars are usually 15% to 35% less expensive than their reference products.
Below are approvals for 2025, including some biosimilars which were the first of their kind.
- In March, Omlyclo® was approved as the first interchangeable biosimilar for Xolair®. Omlyclo manages various allergy and inflammatory conditions.
- In July, Kirsty™ was approved as the first rapid-acting interchangeable biosimilar for NovoLog®. Kirsty helps improve glycemic control in people with diabetes.
- In November, Poherdy® was approved as the first interchangeable biosimilar for Perjeta®. Poherdy can be used in conjunction with other medicines to treat various types of breast cancer.
List of approved biosimilars
References
Biosimilar Product Information. Food & Drug Administration. July 15, 2025.
Avtozma FDA Approval History. Drugs.com. April 30, 2025.
Xbryk FDA Approval History. Drugs.com. February 17, 2025.
Osenvelt FDA Approval History. Drugs.com. July 9, 2025.
Denosumab (subcutaneous route). Mayo Clinic.
Omlyclo FDA Approval History. Drugs.com. March 8, 2025.
Conexxence FDA Approval History. Drugs.com. July 9, 2025.
Jobevne FDA Approval History. Drugs.com. April 15, 2025.
Starjemza FDA Approval History. Drugs.com. June 3, 2025.
Kirsty: Package Insert / Prescribing Info. Drugs.com. September 18, 2025.
Bilprevda FDA Approval History. Drugs.com. December 11, 2025.
Aukelso FDA Approval History. Drugs.com. December 11, 2025.
Enoby FDA Approval History. Drugs.com. December 11, 2025.
Eydenzelt FDA Approval History. Drugs.com. October 12, 2025.
Osvyrti FDA Approval History. Drugs.com. December 11, 2025.
Poherdy FDA Approval History. Drugs.com. November 17, 2025.
Armlupeg FDA Approval History. Drugs.com. November 28, 2025.
LUCENTIS® (ranibizumab). Genentech USA Inc.